Buffers
So how can organisms whose bodies require a near-neutral pH ingest acidic and basic substances (a human drinking orange juice, for example) and survive? Buffers are the key. Buffers, solutions or substances that resist changes in pH readily absorb excess H+ or OH -, keeping the pH of the body carefully maintained in the narrow range required for survival. Maintaining a constant blood pH is critical to a person's well-being. The buffer maintaining the pH of human blood involves carbonic acid (H2CO3), bicarbonate ion (HCO3- ), and carbon dioxide (CO2). When bicarbonate ions combine with free hydrogen ions and become carbonic acid, hydrogen ions are removed, moderating pH changes. Similarly, as shown in the figure below, excess carbonic acid can be converted to carbon dioxide gas and exhaled through the lungs. This prevents too many free hydrogen ions from building up in the blood and dangerously reducing the blood's pH. Likewise, if too much OH - is introduced into the system, carbonic acid will combine with it to create bicarbonate, lowering the pH ( H2CO3 + OH- ![]() HCO3 - + H2O) . Without this buffer system, the body's pH would fluctuate enough to put survival in jeopardy.
HCO3 - + H2O) . Without this buffer system, the body's pH would fluctuate enough to put survival in jeopardy.
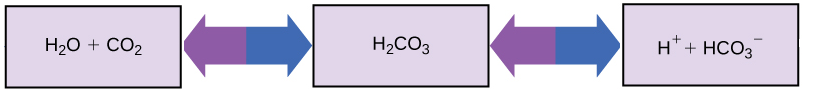
Figure 6. This diagram shows the body's buffering of blood pH levels. The blue arrows represent the process of raising pH as more CO2 is made. The purple arrows indicate the reverse process: the lowering of pH as more bicarbonate is created.
CC BY: Openstax College
Other examples of buffers are antacids used to combat excess stomach acid. Many of these over the-counter medications work in the same way as blood buffers, usually with at least one ion capable of absorbing hydrogen and moderating pH, bringing relief to those that suffer "heartburn" after eating. The unique properties of water that contribute to this capacity to balance pH—as well as water's other characteristics—are essential to sustaining life on Earth.
Self-Assessment



